COVID-19 Vaccine Reduces Long COVID in Children
Published on
Vaccination associated with moderate protection in large, diverse cohort.
Published on
Vaccination associated with moderate protection in large, diverse cohort.
Published on

The Food and Drug Administration (FDA) has approved CASGEVY™ (exagamglogene autotemcel) and LYFGENIA™ (lovotibeglogene autotemcel), the first two gene therapies for the treatment of sickle cell disease in patients 12 years and older with recurrent vaso-occlusive crises (VOCs).
Published on

Alexis Thompson, MD, MPH, received the award at the society’s annual meeting.
Published on
Researchers found CAR T-cell therapy may serve as effective alternative for patients with these disorders.
Published on
CHOP researchers find exposing cells to a small molecule drug improves cell fitness during hematopoietic stem cell transplants, which could improve ex vivo gene therapy.
Published on
Leslie Raffini, MD, and Regina Butler, RN, were quoted in the Philadelphia Inquirer on a story featuring a CHOP patient family living with hemophilia, an inherited bleeding disorder that prevents blood from clotting properly.
Published on

CHOP and Penn researchers have developed a proof-of-concept model for delivering gene editing tools to treat blood disorders directly within the body.
Published on

On a beautiful Saturday in June, Children’s Hospital of Philadelphia (CHOP) hosted the first in-person Cure Sickle Cell Walk & Family Fun Day at Citizens Bank Park raising more than $173,000 to support CHOP’s large and comprehensive sickle cell program.
Published on
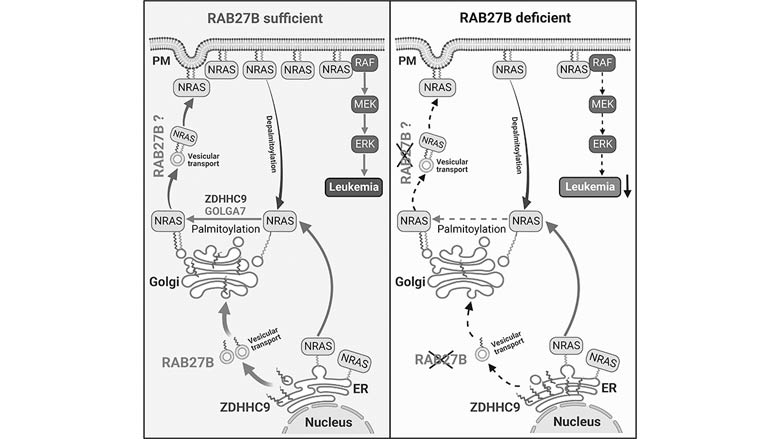
CHOP researchers have identified a molecule that plays a role in post-translational modification and activation in myeloid cancers, making it a potential therapeutic target.
Published on
CHOP gene therapy expert Dr. Lindsey George reacts to a phase 3 hemophilia gene therapy study in an editorial in the New England Journal of Medicine.