Influenza: The Disease & Vaccines
People call it “the flu.” Many people think it is “not that bad.” Some think the vaccine doesn’t work or that it gives them the flu, leading them to opt out of vaccination. Let’s sort out what we know.
Influenza kills thousands to tens of thousands of people every year in the U.S. Often, these deaths do not make headlines, so people don’t realize how many lives this infection takes. Probably the best example of how devastating influenza can be was the influenza pandemic in 1918. This worldwide outbreak killed between 50 million and 100 million people in a single influenza season.
The disease
What is influenza?
Influenza is a virus that infects the windpipe and breathing tubes. Symptoms start suddenly and include high fever, chills, severe muscle aches and headache. The onset of shaking chills is often so dramatic that people will remember the exact hour that it started. Most symptoms only last a few days, but the virus also causes runny nose and a cough that can last for weeks.

On the go? Watch or listen.
Get a summary of the information on this page:
Complications of influenza can include:
- Pneumonia (infection of the lungs)
- Encephalitis (inflammation of the brain)
- Heart disease
- Guillain-Barré syndrome (GBS)
Animals can be infected with influenza
Some diseases are only found in humans; however, influenza can infect many types of animals, including:
- Birds and poultry, such as chickens and turkeys
- Birds that live near water, such as ducks
- Pigs
- Cattle
- Horses
- Dogs
- Cats
- Sea mammals, such as seals and whales
A new type of influenza virus can evolve when animals are infected with different strains of influenza at the same time. The new type of influenza can be dangerous if it has three features:
- It can infect humans.
- It can be easily passed from one person to another.
- It causes people to become ill.
If this happens, a pandemic, or worldwide epidemic, can occur because no one has immunity to the new virus.
Watch this animation to see how influenza virus can change in animals.
What is the difference between an epidemic and a pandemic?
Influenza epidemics occur every year. An epidemic does not affect all people because many people have at least some immunity to the virus.
A pandemic occurs when a new strain emerges because no one has immunity. The result is that virtually everyone is susceptible to infection.
Flu pandemics occur about three times every century. Influenza pandemics have occurred in 1889, 1900, 1918, 1957, 1968, and 2009. The pandemics in 1957 and 1968 each claimed four to six million lives, but the pandemic in 1918 was the most devastating. Between 50 and 100 million people died from the strain of influenza known as "Spanish flu" during that pandemic.
Watch this video clip related to making a vaccine in advance of the arrival of influenza during the fall 1957 pandemic. The clip is from the award-winning documentary, Hilleman: A Perilous Quest to Save the World’s Children.
How does influenza spread?
Influenza spreads through respiratory droplets, such as from coughing and sneezing. It can also spread if someone touches a surface that has the virus on it and then touches their eyes, nose or mouth.
People can also sometimes get influenza after close contact with an infected animal. However, this is not common. Instead, this happens when an animal is infected with a kind of influenza that can also infect people or other animals, such as we heard in the news during 2024 and 2025. At that time, H5N1 influenza virus, a type of influenza common in birds, was found in dairy cattle and a few people.
Find out more about these cases on the Centers for Disease Control and Prevention (CDC) website:
For more information about the history of H5N1 and what to watch for during outbreaks, check the article, “H5N1 Influenza: How to Prevent Fowl Plague from Running Afoul” from our newsletter for science and history enthusiasts, The Hilleman Chronicle.
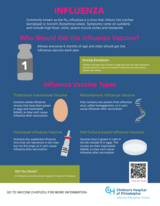
View larger image:
English | Spanish
The vaccine
Who should get the influenza vaccine and when?
Everyone 6 months of age and older is recommended to get the influenza vaccine each year.
Children 6 months to 8 years of age need two doses of influenza vaccine separated by four weeks if they:
- Have never received an influenza vaccine
- Have not received at least two doses of previous influenza vaccines before July 1
Most people should get the influenza vaccine in the fall, preferably September or October. A couple of groups are recommended to get the vaccine earlier (July or August) if it is ready:
- Infants and children getting their first influenza vaccine can get vaccinated earlier since they need two doses separated by a month. This will increase the likelihood that they will be protected before influenza virus starts circulating.
- Pregnant people in their third trimester.
Children of any age who need a single dose can also be vaccinated in the summer, particularly if there is concern that they will not be vaccinated later in the season.
For people who do not get the vaccine in the fall, they can still be vaccinated later in the season because influenza continues to occur in February and March.
Watch this video in which Dr. Paul Offit answers why and when to get the influenza vaccine.
A note about influenza vaccine and egg allergies: Most versions of the influenza vaccine are made in eggs, and some people are severely allergic to eggs, but the small amount of egg proteins in the influenza vaccine do not cause a severe allergic response. For this reason, all versions of the influenza vaccine can be given to people with egg allergies. As is recommended with other vaccines, people should stay at their provider's office for about 15-30 minutes after getting the influenza vaccine.
A note about influenza vaccine and GBS: GBS is a condition in which a person’s immune system mistakenly attacks parts of their nervous system. GBS has been associated with influenza disease and vaccination, so some people (and healthcare providers) think people who have had GBS can’t get the influenza vaccine. However, most people with a history of GBS can actually get this vaccine. The only people with a history of GBS that shouldn’t get the influenza vaccine are those whose GBS occurred within six weeks of receipt of a previous influenza vaccine. In some cases, even these individuals may opt for vaccination after talking with their healthcare provider. For example, they may have other conditions that increase their risk for complications if they get sick with influenza. In these situations, the patient and their healthcare provider should evaluate the potential risks and benefits to decide whether they should get vaccinated. For more information about GBS and vaccines, see this page of our website.
How is the influenza vaccine made?
All of the vaccines listed below contain three influenza strains that circulated in the last year, either in the U.S. or in the Southern hemisphere.
A few different types of influenza vaccine are available:
- One type of influenza vaccine is made by growing influenza viruses in hen's eggs, purifying it, and completely killing it with a chemical (formaldehyde). Historically, this influenza shot has been administered into the muscle.
- One is made using recombinant DNA technology. It contains one protein from the surface of each of the three influenza viruses in the vaccine. The surface protein is called hemagglutinin. This vaccine is given as a shot.
- One is made by growing influenza viruses in mammalian cells (canine kidney cells), instead of eggs. These viruses are killed with the chemical, formaldehyde, so they can’t cause an influenza infection.
- The version of the influenza vaccine given by nose (called intranasal administration) contains live weakened versions of three strains of influenza virus and can be given to people between 2 and 49 years of age. In 2025-2026 an at-home version of this vaccine will be available, so people who qualify can have the vaccine delivered to their home to administer to themselves or others in the home.
For information about ingredients in influenza vaccines, check out the “Flu Vaccine: What’s in the Vial?” infographic (English | Spanish) or accompanying article.
The influenza vaccine is unusual in that most years a different vaccine is made. Because strains of influenza virus that circulate in the community often differ from one season to the next, the vaccine must change to best protect against those different strains. Every year in the U.S., the CDC and the Food and Drug Administration (FDA) work with the World Health Organization (WHO) to determine which strains to include in the vaccine. The influenza vaccine is probably the hardest vaccine to make because of these yearly changes. Hopefully, someday scientists will figure out how to make an influenza vaccine that does not require these yearly updates.
Learn more about why the influenza vaccine changes with each season by watching this short video part of the series Talking About Vaccines with Dr. Paul Offit.
Watch this video in which Dr. Offit answers, “Why Can't We Make a Better Flu Vaccine?”.
Does the influenza vaccine have side effects? Does it cause the flu?
Side effects from the influenza vaccine are very rare. Fever or muscle aches may occur in those who never had influenza or the influenza vaccine before. These symptoms do not mean that a person has "the flu." The vaccine virus is "inactivated" in all but the nasal spray version, so it cannot cause an influenza infection.
The nasal spray version contains live, weakened influenza viruses that can reproduce in the nose, but not the lungs. As such, it can cause mild symptoms, such as runny nose and low-grade fever.
Learn more about the concern that the influenza vaccine can cause influenza in this short video, part of the series Talking About Vaccines with Dr. Paul Offit.
Other questions you might have
What is the difference between winter viruses, colds and "the flu?"
Cold and flu season seems to arrive with winter and the holidays. We see different people, more people, and tend to stay in warm, confined places. All of these things can lead to more sicknesses. Often when people become ill during the winter, they assume they have "the flu." While symptoms for some of these illnesses are "flu-like," the cause may not be influenza. Here is a list of viruses that can cause common winter symptoms:
Respiratory symptoms, like congestion, runny nose, sneezing, coughing, sore throat, can be caused by:
- Influenza virus
- Parainfluenza virus
- Respiratory syncytial virus (RSV)
- Rhinoviruses ("common cold" - over 100 different types)
- Adenoviruses
- Metapneumoviruses
- Human coronaviruses
Gastrointestinal symptoms, like vomiting, nausea, diarrhea, can be caused by:
- Rotaviruses
- Noroviruses
- Enteroviruses (may also cause respiratory symptoms)
- Adenoviruses
- Caliciviruses
- Astroviruses
Only four of the viruses mentioned above (influenza, rotavirus, respiratory syncytial virus (RSV) and one version of coronavirus (SARS-CoV-2, which causes COVID-19)) are currently preventable by immunization.
To lower your chance of getting, or spreading, any of these illnesses:
- Wash your hands frequently and thoroughly.
- Get enough rest.
- Stay home when ill.
- Cough into your elbow.
- Eat a well-balanced diet.
And remember, if you think you have "the flu," you may not actually have influenza.
In this short video, Dr. Lori Handy explains the difference between the common cold and influenza.
Does the influenza vaccine cause GBS?
The data have been mixed regarding whether influenza vaccine can cause GBS; however, it is now generally agreed that very rarely, people can get GBS after receiving the influenza vaccine. However, people can also experience GBS after viral infections, including influenza. Also, the rates of GBS are about 17 times higher after infection than vaccination. For more information, see the September 2021 Parents PACK Feature Article about GBS and vaccines.
Every year, between 75 and 150 previously healthy children die after becoming infected with influenza; therefore, the benefits of getting the influenza vaccine outweigh the risks. The 2024-2025 influenza season was particularly severe, causing around 260 children to die. About 9 of every 10 of these deaths were in children who were not vaccinated against influenza.
When is the best time to get the influenza vaccine?
Most people are recommended to get the influenza vaccine in September or October. Because it takes about two weeks after getting the vaccine to be fully protected, it’s important to get the vaccine before the virus starts spreading in your community.
A few groups of people should be offered the influenza vaccine in August if it is available. These include:
- Women in their third trimester of pregnancy, so their baby can benefit from maternal antibodies before it is old enough to get vaccinated.
- Young children, particularly those who require two doses.
Children of any age who need a single dose can also be vaccinated in the summer, particularly if there is concern that they will not be vaccinated later in the season.
Some people are at increased risk of experiencing complications if they get influenza. Vaccination is particularly important for them. High-risk groups include:
- Young children
- Pregnant women
- Adults 65 years of age and older
- Individuals with underlying medical conditions, such as chronic heart, lung and kidney disease
Why do people still get the flu when they have had the influenza vaccine?
Influenza vaccine protects against the three influenza virus strains expected to circulate during influenza season. However, other factors should be considered.
First, some years the vaccine strains are not as good of a match with the circulating strains as we would like. In these cases, people who were vaccinated may still get influenza. For most of them, their disease will be less severe and shorter than if they had not been vaccinated.
Second, during the period when influenza is common, many other viruses are also circulating. Often these viruses cause colds and other illnesses with similar symptoms, such as congestion, runny nose, sneezing, coughing, sore throat, vomiting, nausea and diarrhea. Sometimes people with these symptoms will think they have influenza even though they are infected with a different virus.
Third, because it takes about two weeks to be protected after getting vaccinated, some people may get the flu before the vaccine response has developed.
Should I get the flu vaccine if I’m pregnant?
Yes. Pregnant women infected with influenza virus are more likely to suffer severe illness and complications that require hospitalization than those of the same age who are not pregnant. Physical changes, including increased blood volume and added stress on the heart and lungs, make influenza infections particularly dangerous during pregnancy. Pregnant women infected with influenza are also at increased risk of premature labor.
Getting an influenza vaccine during pregnancy also provides the newborn with protection for up to six months after birth when he or she is too young to get an influenza vaccine.
- Find out more about infectious diseases and pregnancy.
- Find out more about vaccines during pregnancy: English | Spanish
- Find out more about vaccines and your baby.
What should I do if influenza enters our home?
The influenza vaccine is not perfect, but it’s important to remember that a vaccinated person is likely to have a shorter or less severe illness compared with an unvaccinated person.
Unfortunately, because of low vaccination rates and the imperfect influenza vaccine, vaccinated people might still get the flu. So, what should you know if influenza enters your house?
What to consider at home
Most people with the flu, particularly those who got an influenza vaccination, will have a mild illness that does require medical care or antiviral drugs. If you think you have the flu:
- Get plenty of rest.
- Drink plenty of fluids.
- Return to work or school only after being fever-free for at least 24 hours.
- Stay home — Not only does this help with rest, but it also helps decrease the spread of influenza.
Some people are at greater risk for complications and hospitalization when infected with influenza. They include:
- Children younger than 5 years old — especially those younger than 2 years old.
- Adults older than 65 years of age.
- Children or adults with underlying or chronic conditions of the heart, lungs, endocrine system, kidneys, liver or metabolic system. The CDC offers a comprehensive list of high-risk factors for influenza.
- Children with household members who have compromised immune systems, such as family members receiving chemotherapy for cancer.
- Pregnant women.
Those at higher risk should seek medical care if they think they have influenza. Early medical care may enable treatment with antiviral medications or other specific advice to prevent severe illness, depending on the individual’s situation.
Even though you may be caring for someone with influenza at home, it is important to know the emergency warning signs of influenza illness to watch for based on the person’s age.
Infants
- Being unable to eat
- Trouble breathing
- No tears when crying (a sign of dehydration)
- Significantly fewer wet diapers than normal (another sign of dehydration)
Children
- Fast breathing or trouble breathing
- Bluish skin color
- Not drinking enough fluids
- Not waking up or not interacting
- Being so irritable that they don’t want to be held
- Flu-like symptoms improve but then return with fever and worse cough
- Fever with a rash
Adults
- Difficulty breathing or shortness of breath
- Pain or pressure in the chest or abdomen
- Sudden dizziness
- Confusion
- Severe or persistent vomiting
- Flu-like symptoms that improve but then return with fever and worse cough
What is available if you seek medical care
If you seek medical care, a first step is usually confirming if you have influenza. This is done with a “rapid flu test,” which detects genetic material from the virus. The sample is from a nasal swab, and the results take about 15 minutes.
The test helps healthcare providers determine whether to administer antiviral treatments. Antivirals are most effective when taken within 48 hours of the start of symptoms. If used properly, antivirals may:
- Shorten the length of illness
- Lessen the symptoms
- Prevent those at high risk from developing serious, life-threatening complications
If you seek medical care several days after your illness begins, particularly if you have new or worsening symptoms, healthcare providers will look for complications, like pneumonia, or a secondary infection, meaning an infection with a second type of pathogen that took advantage of your influenza illness. Treatment will depend on your symptoms and whether you have complications or a secondary infection.
Does influenza infection cause people to be more vulnerable to other infections?
Viruses and bacteria sometimes take advantage of the weakened state of a person’s body when they have an infection. When this happens, the person may have infections with two pathogens at the same time or close together in time. The second infection is referred to as a “secondary infection” because it happened as a result of having the first infection. The pathogens that cause the secondary infection are often called “opportunistic.”
One example is that when people have influenza, they are more likely to get a pneumococcal infection. Some other examples of opportunistic pathogens include:
- Group A streptococcus (GAS) — More commonly known as “flesh-eating bacteria,” infects sores caused by chickenpox infection.
- Tuberculosis (TB) — One of the leading causes of death among people living with human immunodeficiency virus (HIV). HIV affects cells of the immune system that make it more difficult to overcome TB infections.
When you are lying in bed with “the flu,” your first thought isn’t that you could get sick with something else. But, influenza infections can make it easier for other infections to follow. Older adults are at greater risk of hospitalization or death following influenza because of these secondary infections. Some will be hospitalized or die from pneumonia caused by influenza. But most will have complications due to pneumococcal infection. If you or someone you know has ever had the flu, seemed to be getting better and then ended up even more ill a few days later, you have witnessed what can happen when a pneumococcal infection follows an influenza infection.
Fortunately, influenza and pneumococcal vaccines are available to protect older adults from this one-two punch.
I heard there is a flu vaccine that can now be ordered to my house. Is that correct?
The flu vaccine administered through the nose and approved for at-home use is called FluMist Home. It was licensed by the FDA in 2024. It is supposed to be available for at-home use starting in the 2025-2026 influenza vaccine season. Currently, only people with private insurance will be able to use this version.
To get the vaccine, a family member will contact the online pharmacy managing the at-home vaccine program and work with them to determine who in the home can get their vaccine this way. You can find out more from our healthcare provider newsletter article, “FluMist at Home.”
Does the flu vaccine contain mercury?
Thimerosal is a mercury-based preservative used to prevent contamination of vaccines in multidose vials. This preservative is no longer used in routinely recommended vaccines used in the U.S., including influenza vaccines. Thimerosal is contained in multidose vials of vaccines used in other countries. Single-dose vials of vaccines do not contain thimerosal.
Two points about thimerosal are important. First the amount of thimerosal in vaccines has been well-studied and found to be safe. Second, this ingredient is important in multi-dose vials of vaccine to prevent harmful contamination. Here’s how: Let’s say the vial contains 10 doses of vaccine. Each dose is removed by putting a needle into the cap on the vial. If the needle or the cap have bacteria on them, those bacteria can get into the vial. So, later doses of vaccine taken from the vial will also contain some bacteria. These bacteria can cause a sore at the injection site. Worse, the bacteria can sometimes get into the bloodstream and cause someone to become very ill. For this reason, making sure vaccines in multi-dose vials contain a preservative, like thimerosal, is important.
Find out more about thimerosal on this page of our website.
Relative risks and benefits
Do the benefits of the influenza vaccine outweigh the risks?
Influenza typically hospitalizes and kills more people in the U.S. than any other vaccine-preventable disease. About 200,000 hospitalizations and thousands to tens of thousands of deaths occur every year. The influenza vaccine causes only mild side effects. Therefore, the benefits of the influenza vaccine clearly outweigh its risks.
Disease risks
- High fever and chills
- Severe muscle aches
- Headaches
- Pneumonia
- Runny nose and coughing for weeks
- Increased risk for secondary infections caused by other pathogens
- Death
Vaccine risks
- Pain, redness and swelling at the injection site
- Fever or muscle aches
References
Orenstein W, Offit PA, Edwards KM and Plotkin SA. Inactivated and Recombinant influenza vaccines in Plotkin’s Vaccines, 8th Edition. 2024, 514-551.
Orenstein W, Offit PA, Edwards KM, and Plotkin SA. Influenza vaccine — Live in Plotkin’s Vaccines, 8th Edition. 2024, 552-576.
Reviewed by Paul A. Offit, MD, on August 14, 2025
