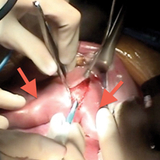
Teratomas are tumors derived from a combination of multiple germinal cell layers. Massive growth of a teratoma with expansion of tumor size can impede functionality of adjacent structures. Rapid growth of a sacrococcygeal teratoma can be associated with increased vascularity, which may lead to cardiovascular volume overload and heart failure in utero. Teratomas within the chest can behave somewhat differently. These tumors can impinge upon the esophagus or airway, resulting in changes in amniotic fluid, or cause critical airway obstruction at birth. If large, teratomas within the chest can impede filling of the heart, leading to decreased cardiac output (1).
Pericardial teratomas are rare tumors that arise from the sac lining of the heart. They can grow rapidly, leading to pericardial effusion and an in utero cardiac tamponade physiology. Massive growth of the tumor compresses the heart, leading to fetal hydrops and death. When identified after birth, resection is relatively straightforward and can be lifesaving. However, when identified in fetal life, complex decisions must be made. The goal of prolongation of the pregnancy to as mature a gestation as possible must be balanced against the competing process of rapidly progressive tumor growth with impending hydrops and fetal demise. If a rapidly growing pericardial teratoma is seen early in gestation, management options are few and outcome is usually poor.
Case study
A 23-year-old woman, gravida two, para one, Vermont native presented locally at 20 weeks’ gestation for routine obstetrical ultrasound examination. A chest mass was identified with suspicion of pericardial teratoma. The patient was referred to the Center for Fetal Diagnosis and Treatment and Fetal Heart Program at The Children’s Hospital of Philadelphia for further evaluation. Ultrasound and fetal echocardiographic imaging performed at 22 weeks' and 6 days' gestation revealed a large pericardial teratoma. It originated from the pericardial reflection over the right atrium-right ventricular groove, and was draped over the arising ascending aorta. The tumor was amorphous, with heterogeneous echo-texture and a number of small cysts. The mass compressed the right-sided cardiac structures. There was a small pericardial effusion, but no overt hydrops.
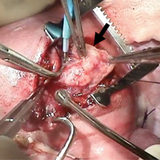
Recommendation was made for follow-up assessment in one week’s time to gauge rate of tumor growth. Follow-up study revealed profound, rapid growth compared to one week prior, with the tumor measuring 2.5 x 3 cm in size. The mass was now markedly compressing the right atrium and impinging upon the ascending aorta, causing flow disturbance. There was decreased cardiac output with the Doppler-derived measurement at approximately 300 cc/kg/ min, normal range being 400–500 cc/kg/min. Pericardial effusion was now moderate in size.
Increased tumor growth, compression of the right side of the heart, and decreased cardiac output at this gestational age suggested the fetus would likely not survive much longer in utero, let alone make it to term. The seriousness of the situation was discussed with the parents and the possibility of fetal intervention was raised. Open fetal surgery for resection of pericardial teratoma had been previously described in a hydropic fetus; however, to date there were no reports of survivors (2). After extensive counseling concerning the risks and potential benefits, the family chose to have open fetal surgery with resection of the teratoma.

At 24 weeks’ gestation, a multidisciplinary team consisting of CHOP experts in fetal surgery, cardiothoracic surgery, maternal-fetal medicine, fetal cardiology, anesthesia and nursing performed the operation. Using fetal surgical techniques previously established at CHOP for other intrathoracic anomalies, the uterus was opened and the fetal chest exposed. A median sternotomy was performed and a giant pericardial teratoma was carefully dissected and removed off the beating heart. Continuous echocardiographic imaging during the procedure allowed for cardiovascular monitoring and helped guide the conduct of the operation (3, 4). As the tumor was slowly removed, volume loading with simultaneous administration of blood under direct echo inspection reduced the risk of cardiovascular collapse, which can occur with sudden relief of cardiac tamponade. The fetal sternum and maternal uterus were closed. Both fetus and mother tolerated the operation well. The pregnancy was monitored carefully with serial evaluation, and there was no evidence of return of the tumor. A healthy infant was delivered via C-section at 38 weeks’ gestation, without any evidence of cardiovascular problems.
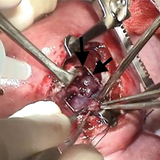
Thoughtful incorporation of multidisciplinary input from multiple experts allowed for correct judgments to be made concerning the proper timing to alter the natural history and intervene surgically in this case, leading to an excellent result.
References
- Merchant AM, Hedrick HL, Johnson MP, Wilson RD, Crombleholme TM, Howell LJ, Adzick NS, Flake AW. Management of fetal mediastinal teratoma. J Pediatr Surg. 2005 Jan;40(1):228–31.
- Sydorak RM, Kelly T, Feldstein VA, Sandberg PL, Silverman NH, Harrison MR, Albanese CT. Prenatal resection of a fetal pericardial teratoma. Fetal Diagn Ther. 2002 Sep–Oct; 17(5):281–5.
- Rychik J, Tian Z, Cohen MS, Ewing SG, Cohen D, Howell LJ, Wilson RD, Johnson MP, Hedrick HL, Flake AW, Crombleholme TM, Adzick NS. Acute cardiovascular effects of fetal surgery in the human. Circulation. 2004 Sep 21;110(12):1549–56.
- Keswani SG, Crombleholme TM, Rychik J, Tian Z, Mackenzie TC, Johnson MP, Wilson RD, Flake AW, Hedrick HL, Howell LJ, Adzick NS. Impact of continuous intraoperative monitoring on outcomes in open fetal surgery. Fetal Diagn Ther. 2005 Jul–Aug;20(4):316–20.
Featured in this article
Specialties & Programs

Teratomas are tumors derived from a combination of multiple germinal cell layers. Massive growth of a teratoma with expansion of tumor size can impede functionality of adjacent structures. Rapid growth of a sacrococcygeal teratoma can be associated with increased vascularity, which may lead to cardiovascular volume overload and heart failure in utero. Teratomas within the chest can behave somewhat differently. These tumors can impinge upon the esophagus or airway, resulting in changes in amniotic fluid, or cause critical airway obstruction at birth. If large, teratomas within the chest can impede filling of the heart, leading to decreased cardiac output (1).
Pericardial teratomas are rare tumors that arise from the sac lining of the heart. They can grow rapidly, leading to pericardial effusion and an in utero cardiac tamponade physiology. Massive growth of the tumor compresses the heart, leading to fetal hydrops and death. When identified after birth, resection is relatively straightforward and can be lifesaving. However, when identified in fetal life, complex decisions must be made. The goal of prolongation of the pregnancy to as mature a gestation as possible must be balanced against the competing process of rapidly progressive tumor growth with impending hydrops and fetal demise. If a rapidly growing pericardial teratoma is seen early in gestation, management options are few and outcome is usually poor.
Case study
A 23-year-old woman, gravida two, para one, Vermont native presented locally at 20 weeks’ gestation for routine obstetrical ultrasound examination. A chest mass was identified with suspicion of pericardial teratoma. The patient was referred to the Center for Fetal Diagnosis and Treatment and Fetal Heart Program at The Children’s Hospital of Philadelphia for further evaluation. Ultrasound and fetal echocardiographic imaging performed at 22 weeks' and 6 days' gestation revealed a large pericardial teratoma. It originated from the pericardial reflection over the right atrium-right ventricular groove, and was draped over the arising ascending aorta. The tumor was amorphous, with heterogeneous echo-texture and a number of small cysts. The mass compressed the right-sided cardiac structures. There was a small pericardial effusion, but no overt hydrops.

Recommendation was made for follow-up assessment in one week’s time to gauge rate of tumor growth. Follow-up study revealed profound, rapid growth compared to one week prior, with the tumor measuring 2.5 x 3 cm in size. The mass was now markedly compressing the right atrium and impinging upon the ascending aorta, causing flow disturbance. There was decreased cardiac output with the Doppler-derived measurement at approximately 300 cc/kg/ min, normal range being 400–500 cc/kg/min. Pericardial effusion was now moderate in size.
Increased tumor growth, compression of the right side of the heart, and decreased cardiac output at this gestational age suggested the fetus would likely not survive much longer in utero, let alone make it to term. The seriousness of the situation was discussed with the parents and the possibility of fetal intervention was raised. Open fetal surgery for resection of pericardial teratoma had been previously described in a hydropic fetus; however, to date there were no reports of survivors (2). After extensive counseling concerning the risks and potential benefits, the family chose to have open fetal surgery with resection of the teratoma.

At 24 weeks’ gestation, a multidisciplinary team consisting of CHOP experts in fetal surgery, cardiothoracic surgery, maternal-fetal medicine, fetal cardiology, anesthesia and nursing performed the operation. Using fetal surgical techniques previously established at CHOP for other intrathoracic anomalies, the uterus was opened and the fetal chest exposed. A median sternotomy was performed and a giant pericardial teratoma was carefully dissected and removed off the beating heart. Continuous echocardiographic imaging during the procedure allowed for cardiovascular monitoring and helped guide the conduct of the operation (3, 4). As the tumor was slowly removed, volume loading with simultaneous administration of blood under direct echo inspection reduced the risk of cardiovascular collapse, which can occur with sudden relief of cardiac tamponade. The fetal sternum and maternal uterus were closed. Both fetus and mother tolerated the operation well. The pregnancy was monitored carefully with serial evaluation, and there was no evidence of return of the tumor. A healthy infant was delivered via C-section at 38 weeks’ gestation, without any evidence of cardiovascular problems.

Thoughtful incorporation of multidisciplinary input from multiple experts allowed for correct judgments to be made concerning the proper timing to alter the natural history and intervene surgically in this case, leading to an excellent result.
References
- Merchant AM, Hedrick HL, Johnson MP, Wilson RD, Crombleholme TM, Howell LJ, Adzick NS, Flake AW. Management of fetal mediastinal teratoma. J Pediatr Surg. 2005 Jan;40(1):228–31.
- Sydorak RM, Kelly T, Feldstein VA, Sandberg PL, Silverman NH, Harrison MR, Albanese CT. Prenatal resection of a fetal pericardial teratoma. Fetal Diagn Ther. 2002 Sep–Oct; 17(5):281–5.
- Rychik J, Tian Z, Cohen MS, Ewing SG, Cohen D, Howell LJ, Wilson RD, Johnson MP, Hedrick HL, Flake AW, Crombleholme TM, Adzick NS. Acute cardiovascular effects of fetal surgery in the human. Circulation. 2004 Sep 21;110(12):1549–56.
- Keswani SG, Crombleholme TM, Rychik J, Tian Z, Mackenzie TC, Johnson MP, Wilson RD, Flake AW, Hedrick HL, Howell LJ, Adzick NS. Impact of continuous intraoperative monitoring on outcomes in open fetal surgery. Fetal Diagn Ther. 2005 Jul–Aug;20(4):316–20.
Contact us
Fetal Heart Program