CAR T-Cell Therapy
CAR T-cell therapy is a new approach to cancer treatment in which we use the bodies’ own immune cells, which fight infection, to kill off cancer cells.
Cancer researchers have spent decades studying immunotherapy as a possible treatment for cancer. Over the years they solved many of the problems associated with this type of treatment, including learning how to:
- Teach the cells to recognize cancer
- Grow cells in a lab in way that it would be safe to re-infuse them into patients
- Program a cell that would continue to grow in the body for more than a few days, and then remain in the body for an extended period of time to continue controlling a patient’s disease for the long-term
The form of CAR T-cell therapy developed through a collaboration of Children’s Hospital of Philadelphia and the Hospital of the University of Pennsylvania solves all of these problems, and was FDA approved for use in children with relapsed B-cell acute lymphoblastic leukemia in 2017.
How does CAR T-cell therapy (immunotherapy) work?
In this cancer treatment, immune cells called T cells are taken from a patient's own blood. These cells are genetically modified to express a protein which will recognize and bind to a target called CD19, which is found on cancerous B cells.
T cells, a type of white blood cell, are designed to kill disease cells. Cancerous B cells often look like your own normal, healthy cells, so T cells don’t go after them.
We collect millions of T cells from the patient, then we reprogram them in a lab so they can now identify the cancerous B cells and grab onto a substance that is found only on the surface of B cells.
When we put the reprogrammed T cells back into the patient they flow throughout the body and begin locating cancerous B cells.
As the reprogrammed T cells attach to and destroy the rapidly dividing cancerous B cells, they also multiply in the body and can remain in the body long after to continue fighting any new cancerous B cells. They will also kill the healthy B cells. A patient who undergoes this therapy will not have any B cells in their immune system.
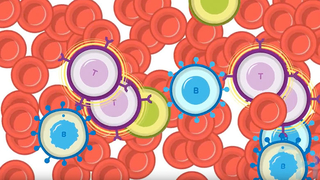
Image: CAR T cells attach to and destroy rapidly dividing cancerous B cells.
Who might benefit from CAR T-cell therapy?
CAR T-cell therapy is approved for treatment of children and adolescents with fairly advanced B cell acute lymphoblastic leukemia (ALL), but not other leukemias or pediatric cancers. It is an option for those patients who have very resistant ALL. Research is ongoing to extend this therapy to other types of pediatric cancer.
Roughly 85 percent of ALL cases are treated very successfully with standard chemotherapy. For the remaining 15 percent of cases, representing a substantial number of children in the United States, chemotherapy only works temporarily or not at all. This is not a treatment for newly diagnosed leukemia, only for patients whose leukemia is not responding to chemotherapy, and whose disease has come back after a bone marrow transplant.
What are the stops in the CAR T-cell therapy process?
There are four main stops in the T-cell therapy process:
1. Your first stop will be on the third floor of the Buerger Center for Advanced Pediatric Care. You’ll meet with a Cancer Immunotherapy team member who will take labs and talk to your child and family about the care plan. Then you’ll take a tour of our facilities.
2. Next, you’ll be in CHOP’s Main Hospital, across the street from the Buerger Center. Our team of interventional radiologists will place a central line for apheresis.
3. The third stop is apheresis, which is done on the second floor of the Buerger Center. Every child treated with T-cell therapy will have cells collected through this process. The collected cells are used to create the personalized treatment — called “CAR T-cell product.”
4. The last stop is the infusion of reprogrammed T cells. The infusion typically happens during another, longer visit to CHOP. Here’s what will happen during that visit:
- Chemotherapy for disease control; a baseline disease evaluation
- Infusion and monitoring; this happens in the Day Hospital on the 3rd floor of the Buerger Center. The therapy is given by injection of the cells into your central line. It only takes a few minutes.
- A period of regular clinic visits for monitoring and laboratory testing
- Response assessment: diagnostic lumbar puncture, bone marrow aspirate, biopsy
- Return home
We will continue to follow your child closely after your initial stay in Philadelphia. Some visits will happen at home, and some visits will take place at CHOP.
Monitoring for side effects of CAR T-cell therapy
During and after the immunotherapy infusion we will closely monitor your child for side effects. If your child develops a fever we will use standard fever management and they will be admitted to our oncology unit in the Main Hospital.
Some children develop more serious side effects requiring transfer to our Pediatric Intensive Care Unit (PICU). Our PICU staff is especially equipped to manage serious side effects, since our Cancer Immunotherapy team developed the care plans here that are now used worldwide.
Referring a patient for CAR T-cell therapy
Referring physicians and families are encouraged to consider the potential benefits of cell therapy early in the cancer treatment process. In the case of CAR T-cell therapy, it is critical that cells are collected from the patient before they are too sick. Also, intensive chemotherapy will decrease the number of normal T cells we need to collect from the patient.
To refer a patient, call 267-426-0762 or contact our Cancer Center online. Learn more about what physicians and families can expect throughout the referral process.