Vertical expandable prosthetic titanium rib (VEPTR)
The vertical expandable prosthetic titanium rib (VEPTR) is the only FDA-approved device to treat children with thoracic insufficiency syndrome, a congenital condition where severe deformities of the chest, spine and ribs prevent normal breathing, lung growth and lung development.
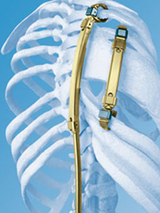
VEPTR is a curved metal rod that is surgically attached to a child’s ribs, spine or pelvis using hooks on both ends of the device. The VEPTR helps straighten the child’s spine and separate ribs so their lungs can grow and expand as the child grows.
The length of the VEPTR device can be expanded or contracted as the patient grows.
Creating the VEPTR
Robert M. Campbell Jr., MD, director of the Wyss/Campbell Center for Thoracic Insufficiency Syndrome at Children’s Hospital of Philadelphia (CHOP), designed the world’s first expandable rib in 1988. The original designs led to blue prints and official plans with Techmedica, a custom prosthesis firm in California.
Campbell and Melvin Smith, MD, a pediatric general surgeon, implanted the first titanium rib into a Texas child on April 19, 1989. As their experience with VEPTR grew and more critically ill children were implanted with the device, the device was modified.
In 1994, Synthes Spine Co., part of Synthes, Inc., an international company based in West Chester, PA, took over production of the VEPTR, and has been producing it ever since.
On Aug. 23, 2004, the U.S. Food and Drug Administration approved VEPTR (H030009) as a medical device under the Humanitarian Device Exemption Program. A Humanitarian Use Device is intended to treat or diagnose a disease or condition — such as thoracic insufficiency syndrome — that affects fewer than 4,000 people in the U.S. per year.
Is VEPTR the right treatment for your child?
When you bring your child to the Wyss/Campbell Center for Thoracic Insufficiency Syndrome at CHOP, we will fully evaluate your child with a combination of tests and consultations with our multidisciplinary team of specialists from orthopedics, general surgery, radiology, pulmonology, neurology, genetics and anesthesia.
If your child is diagnosed with thoracic insufficiency syndrome, VEPTR treatment is generally recommended. If your child has a spine disorder, but no related lung or chest issues, VEPTR is generally not an appropriate treatment option.
Instead, doctors may recommend other surgical or nonsurgical options to address your child’s condition, such as:
- Back bracing — the most common nonsurgical treatment for scoliosis, kyphosis and other congenital spine conditions
- Growing rods — the most common surgical treatment for growing children (who have not reached adult height) with significant spine curvature due to scoliosis, kyphosis and other congenital spine disorders
- Spinal fusion — the most common surgical treatment for children (who have reached skeletal maturity) with significant spine curvature due to scoliosis, kyphosis and other congenital spine disorders
Learn more about thoracic insufficiency syndrome and related congenital conditions.
How VEPTR works
If doctors recommend VEPTR surgery for your child — and you agree that is the best option — we will schedule pre-op testing and surgery. In most cases, children receive their initial implant surgery before age 5.
During surgery
During surgery, doctors attach and adjust the VEPTR device to fit your child. The device is generally attached vertically on your child’s ribs near the spine. In some cases, more than one VEPTR device is used to create space in the chest for the lungs to develop more fully. VEPTRs can also be attached to the spine or pelvis, depending on your child’s specific anatomy.
Recovery at home after surgery
After your child is discharged from the hospital, they will return home to continue healing. About three weeks after surgery, we recommend that patients follow up with either our team or their primary care provider for an incision check.
If you are concerned about your child’s wound – especially if you notice fluid draining from the incision or your child develops a fever – please contact your child’s orthopedic team at 215-590-1527.
We may ask you to take pictures of your child’s wound and send them electronically for evaluation by our team.
Follow-up care
As your child grows, they will need to return for check-ups and to schedule VEPTR adjustment surgery as needed. Additional follow-up will be determined by your child’s clinical team but is typically every 6-9 months until your child reaches skeletal maturity (usually between age 10 and 16).
Because your child is growing rapidly during these years, surgery will need to be done to expand the VEPTR device, growing rods or combination of the two to ensure your child’s lungs also continue growing and keep pace with their development. These adjustments allow your child to continue to grow taller, as well as further correct spine and chest wall deformities. These adjustments are made through a small incision in your child's back.
In most cases, these additional surgeries will require one overnight stay in the hospital and recovery time is far quicker than the initial implantation surgery.
Treatment results
Treatment results with any surgical procedure can vary from child to child. In general, VEPTR treatment results in:
- A more normal growth pattern
- Decreased chest, spine and rib deformities
- Decreased need for supplemental oxygen
- Expanded lung volume
- Increased life span
- Increased physical activity capability
- Improved psychosocial health
Growing use of VEPTR treatment
In the past 20 years, thousands of VEPTRs have been implanted into children in more than 25 countries worldwide.
CHOP has been performing VEPTR surgery on children with thoracic insufficiency syndrome for nearly 10 years and has performed hundreds of VEPTR implant and revision surgeries.
Last year alone, our team treated more than 200 children with thoracic insufficiency syndrome with VEPTR implants/expansions. Some children need two VEPTR devices to correct their chest wall or spine anomalies. In rare cases, three VEPTRs are used.
Long-term outcomes
Long-term outcomes for children undergoing VEPTR surgery are generally good. The VEPTR allows your child’s lungs to grow as they grow.
When your child reaches skeletal maturity, our team will likely recommend spinal fusion surgery to make the spine and chest wall correction permanent.
If your child has missing or fused ribs, we may recommend your child keep a VEPTR device into adulthood indefinitely. Your child — and the device — will need to continue to be monitored, but the VEPTR will continue to act as a replacement rib, ensuring your child’s chest wall remains open, lung volume is maximized, and internal organs are protected.
Ongoing research
Our Center continues to research VEPTR treatment, infection rates and long-term outcomes for children with thoracic insufficiency syndrome.
More experience is needed to provide more complete answers about long-term outcomes for children with thoracic insufficiency syndrome. Learn more about our research and our safety registry.