The World’s Smallest Pacemaker: Cameron’s Story
The World’s Smallest Pacemaker: Cameron’s Story
When pregnant mom, Shakiara, learned that her lupus placed her unborn child at a higher risk of congenital complete heart block, she began to receive weekly fetal checks with her care team at the Hospital of the University of Pennsylvania (HUP). Her baby presented well until 21 weeks, when the exam indicated signs of heart block. Within hours, Shakiara was seen in the Fetal Heart Program at Children’s Hospital of Philadelphia (CHOP).
“The connection between the Fetal Heart Program and HUP was seamless,” says Shakiara. “Their collaboration was amazing, which really helped relieve my stress.”
A fetal echocardiogram confirmed the diagnosis of heart block, a condition that occurs when the electrical impulses that control the heartbeat are disrupted.
While healthy mothers in the Fetal Heart Program have the option of delivering in the Garbose Family Special Delivery Unit, Shakiara was sick and needed the care of her HUP team. “It was important to me that they cared about my safety as well as my baby’s,” she says.
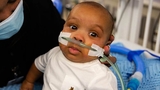
On Nov. 23, 2022, Cameron was born at 34 weeks at HUP and immediately transferred to CHOP’s Cardiac Intensive Care Unit (CICU).
A collaborative approach
In most cases, children born with heart block will eventually require a pacemaker as their heart rate is far below a typical newborn’s rate of 130 to 150 beats per minute. However, patients without symptoms and a heart rate in the mid-50’s may not require pacemaker intervention. Cameron’s heart rate hovered in the low 50’s.
“We try not to pace unless needed,” says, Maully J. Shah, MBBS, FACC, FHRS, CCDS, CEPS, Medical Director of CHOP’s Electrophysiology and Heart Rhythm Program. “A pacemaker can sometimes cause complications in small babies, and we were hoping that by not imposing another procedure on Cameron, he would continue to progress on his own.”
In addition to heart block, Cameron was also born with pulmonary hypertension and tachypnea, abnormally rapid breathing that can make it unsafe for infants to feed orally. When Cameron’s breathing and feeding issues failed to improve, his NICU team felt a higher heart rate might help him cope with these additional medical challenges. The neonatal, pulmonary and electrophysiology teams consulted and agreed: a pacemaker was the right approach.
Says Dr. Shah, “We began to think about how we could give him a pacemaker with the least potential for complications.”
‘The size of a triple A battery’
A conventional pacemaker weighs approximately 27 grams (nearly one ounce), and the additional weight could potentially make Cameron’s breathing and feeding problems worse. The smallest commercially available pacemaker — Medtronic’s Micra — weighs only two grams, but is a leadless pacemaker designed to be delivered by catheter to the heart.
“We thought the Micra could be adapted and implanted surgically as an epicardial pacemaker,” says Dr. Shah. An epicardial pacemaker is connected to the heart by leads — wires that run from the pulse generator to the heart. Using 3-D printing, Medtronic converted the leadless pacemaker to a pacemaker with an adaptor to attach a lead. “We got [the adapted Micra] down to two grams, about the size of a triple A battery.”
The lower weight of the adapted pacemaker, also known as an implantable pulse generator (IPG), would be less likely to complicate Cameron’s breathing and feeding issues. In addition, the tiny pacemaker could now be implanted through a smaller incision, which would enable a faster recovery and allow Cameron’s care team to concentrate on his additional medical issues.
Cameron would be one of only 10 babies in the world to receive the IPG, and the first ever at CHOP.
The only problem? While the Micra is FDA-approved, this adapted version is not.
A difficult decision
While pacemakers are frequently used in adults, they are less commonly used in children, especially babies. Only about 1 in 20,000 children are born with heart block each year, and of those children, not all of them require a pacemaker. As such, there is very little demand for pacemakers smaller than the Micra.
“There are many obstacles to the IPG becoming commercially available,” says Dr. Shah. “While it’s not profitable to manufacture due to the low demand, the pediatric population is vulnerable. The industry and the FDA should develop avenues to manufacture devices that serve this population.”
Before Cameron could receive the pacemaker, it would need to be approved by the FDA for investigational use on humanitarian grounds. This would involve a mountain of paperwork — and, above all, Shakiara needed to be comfortable with the plan.
“I was worried,” she admits. “My reassurance came when Dr. Shah said that it’s worked in other children. I trust her, and the fact that she collaborates with doctors across the country gave me a lot of reassurance that this was the best decision.”
Preparing for surgery
With Shakiara on board and approval underway, the team began to plan for Cameron’s surgery.
“The goal was to minimize the footprint of the device,” says Constantine D. Mavroudis, MD, MSc, MTR, an attending cardiothoracic surgeon. “It wasn’t until I held the device in my hand that I realized what I could really do with it. I was happy to take the challenge.”
A larger pacemaker would require a longer incision and create a greater opportunity for wound complications, such as infections. The size of the IPG enabled Dr. Mavroudis to take a minimally invasive approach with only a 2.5 cm incision (less than one inch) — as opposed to the standard 9 cm (3.5 inches) — and keep Cameron’s sternum fully intact.
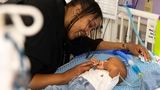
Says Shakiara: “Cameron’s surgical site is beautiful. When you think about how small his body is, the fact that his scar isn’t noticeable is amazing.”
The road to home
After surgery, Cameron remained in the NICU where the nurses, who don’t typically care for preemies with pacemakers, received special training about how pacemakers work and what to expect.
“From the get-go, the [electrophysiology] team was here educating me and answering all my questions,” says Shakiara.
Once Cameron is discharged home, the pacemaker will — through a home transmitter — automatically transmit data to Cameron’s care team, enabling them to monitor how well the device is working. He will likely need another pacemaker in 5-7 years.
In the meantime, Cameron has also received a g-tube for feeding and his care team continues to focus on lowering his respiratory rate so that his breathing can be managed at home.
“Now that the pacemaker is implanted,” says Shakiara, “we’re working our way home.”