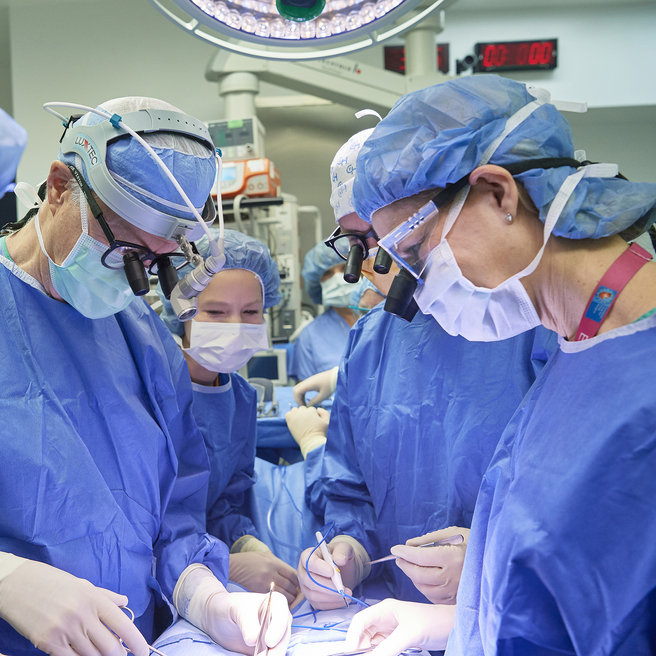
What is twin-twin transfusion syndrome (TTTS)
Twin-twin transfusion syndrome (also called TTTS or twin to twin transfusion syndrome) is a condition in which the blood flows unequally between twins that share a placenta (monochorionic twins). TTTS occurs in about 10 to 15 percent of monochorionic, diamniotic (two amniotic sacs) twins.
What causes TTTS?
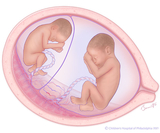
In twin-twin transfusion syndrome, there is an unequal sharing of blood that passes between twins through blood vessel connections on the surface of the placenta. One twin (called the donor twin) pumps blood to the other twin (called the recipient). This causes the recipient twin to receive too much blood and the donor twin to receive too little.
There are no known genetic causes of TTTS.
What are the symptoms of TTTS?
In TTTS, the increased volume of blood causes the recipient twin to produce more than the usual amount of urine, which can result in a large bladder, too much amniotic fluid (known as polyhydramnios) and hydrops, a prenatal form of heart failure.
The donor twin, who receives too little blood, produces less than the usual amount of urine. This results in low or no amniotic fluid surrounding it (oligohydramnios) and a small or absent bladder.
Without intervention, the condition can be fatal for both twins.
Women carrying babies with TTTS may experience abdominal discomfort from polyhydramnios, and if left untreated, potential rupture of membranes and premature labor.
Evaluation and diagnosis of TTTS
The most important step in evaluating a twin pregnancy for twin-twin transfusion syndrome is to determine whether the twins share a single placenta. An ultrasound examination early in pregnancy (during the first trimester) can best make this distinction.
Other features of TTTS include twins of the same gender, a difference in size between the twins, and a difference in the volume of amniotic fluid surrounding the two fetuses. A detailed sonographic assessment rules out any additional structural anomalies.
Twin-twin transfusion syndrome can also have a huge impact on the cardiovascular system in twins. It is important that twins with TTTS are thoroughly evaluated for heart problems while in utero.
If you are referred to CHOP’s Center for Fetal Diagnosis and Treatment, you will be scheduled for a comprehensive, one-day evaluation. During this first visit, you will meet with multiple team members, including a genetic counselor, nurse coordinator and maternal-fetal medicine specialist. You’ll also meet with a research coordinator to discuss ongoing research studies.
Your evaluation will involve the following exams:
- High-resolution ultrasound – An examination to rule out structural abnormalities in the twins. This exam will also confirm placental location and umbilical cord insertions into the placenta, assess amniotic fluid levels in each sac and assess blood flow patterns in specific blood vessels for each twin.
- Fetal echocardiogram – A focused ultrasound of each twin’s heart to look for any related heart conditions. Twin-twin transfusion syndrome may cause serious cardiac strain in the recipient twin, resulting in enlargement of the heart and cardiac dysfunction. All twins evaluated for TTTS should undergo a fetal echocardiogram. Fetal cardiology experts from CHOP’s Fetal Heart Program have developed a cardiac scoring tool to determine how severe the condition has become for the twins. This score helps the medical team determine need for treatment. (See “The twin-twin transfusion syndrome: Spectrum of cardiovascular abnormality and development of a cardiovascular score to assess severity of disease.”)
- Genetic amniocentesis – A procedure in which a small amount of amniotic fluid is removed from the sacs surrounding the fetuses and tested. In some cases, this genetic test may be needed if imaging studies identify structural malformations in addition to TTTS.
Accurate diagnosis is extremely important in distinguishing TTTS from other related anomalies, such as selective intrauterine growth restriction (sIUGR), also called selective fetal growth restriction (sFGR). TTTS and sIUGR/sFGR can both occur in monochorionic, diamniotic pregnancies, but are differentiated by the lack of polyhydramnios/oligohydramnios in sIUGR/sFGR. The management of the two conditions are different, and therefore it is important to have a thorough evaluation to determine the most accurate diagnosis.
Your test results and specific treatment options will be discussed in detail on the day of your evaluation. If fetal intervention is recommended, you can also expect to meet with an advanced practice nurse to complete preoperative testing, and a social worker to arrange temporary accommodations in the Philadelphia area. Our team is here to help and support you and your family throughout this experience.
(Healthcare professionals: for more diagnostic tools and resources to help in the management of patients with complicated twin pregnancies, please refer to our monochorionic pregnancy resources for clinicians.)
Stages of TTTS
In TTTS, there is a characteristic series of pregnancy changes that happens due to the extra blood coming from the donor twin to the recipient twin.
Stage 1
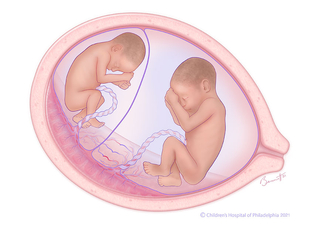
In TTTS, unequal blood flow between twins sharing a placenta results in recipient twin (right) having too much amniotic fluid (polyhydramnios), and donor twin (left) with little or no amniotic fluid (oligohydramnios). This illustration depicts stage 1 TTTS.
Stage 2
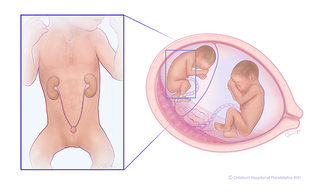
In stage 2 TTTS, the donor twin’s bladder (left) is not filling or emptying, because blood is being diverted from the kidneys to the brain, heart, and adrenal glands. This is a survival mode mechanism to preserve the functioning of these vital organs. The kidneys and bladder are still functioning, but more slowly than normal, so you don't see the bladder filling and emptying as normal.
Stage 3
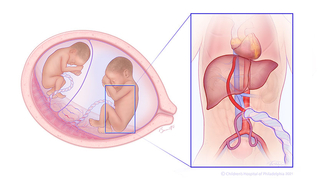
In stage 3 TTTS, abnormal blood flow patterns include reversed flow in ductus venosus (DV), and absent/reversed umbilical artery end-diastolic velocity (UAEDV). Abnormal dopplers (blood flow patterns) indicate the heart function of one or both twins is affected.
Stage 4
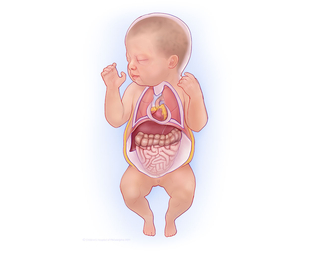
In stage 4 TTTS, hydrops (massive fluid retention) develops, which puts both mother and twins at risk. Hydrops is indicative of heart failure in the recipient twin. The recipient's heart is struggling to process all the extra blood being sent from the donor twin.
Stage 5
In stage 5 TTTS, death of one or both twins while still in the uterus can occur.
Twin-twin transfusion syndrome treatment
Treatment for twin-twin transfusion syndrome may include any of the following:
- Expectant management – In situations where surgery is not yet indicated (Stage 1 without additional risk factors), close monitoring with periodic ultrasound examinations is used to evaluate the condition of both twins and look for signs of progression. In some cases, a follow-up fetal echocardiogram is used as well to look for signs of cardiac changes; these may sometimes be seen before other changes.
- Fetoscopic selective laser ablation – A minimally invasive surgery performed on the placenta to disconnect the communicating blood vessels. This procedure, also called selective laser photocoagulation (SLPC), stops the sharing of blood from the donor to the recipient, with the goal of halting the progression and then resolving the twin-twin transfusion syndrome. This is typically the preferred treatment for TTTS, depending on gestational age at presentation, location of the placenta and stage of TTTS.
- Amnioreduction – Removal of excess amniotic fluid from the larger twin (recipient) which may help ease any pain or discomfort experienced by the mother due to fluid buildup. This is a temporary treatment option and may need to be repeated.
- Selective cord occlusion – A minimally invasive surgery that stops the blood flow to one twin in order to maximize the outcome for the other twin. Selective cord occlusion procedures include radiofrequency ablation (RFA) and bipolar cord coagulation (BCC). This is considered a last resort option when the disease is very advanced and the at-risk twin is not going to survive. This intervention can protect the co-twin from neurologic impairment and/or death.
Mothers undergoing a fetal surgery procedure will stay in our Garbose Family Special Delivery Unit, the first birthing unit within a pediatric hospital dedicated to healthy mothers carrying babies with serious and life-threatening birth defects.
Watch our educational video series to learn more about the diagnosis and treatment of twin-to-twin transfusion syndrome. En Español »
Follow-up care for TTTS
If you undergo fetal intervention, your care team will provide detailed postoperative care and instructions. We will schedule you for an ultrasound exam at our Center one week after your procedure to re-evaluate the health of your twins.
After that exam, we recommend returning to your local maternal-fetal medicine specialist for weekly ultrasound examinations for at least three weeks. Ultrasound exams will then be scheduled according to your doctor for the duration of your pregnancy.
Volumes & outcomes
When seeking the best hospital for TTTS treatment, we encourage you to ask about any treatment team about their volumes and outcomes. Our team at the Center for Fetal Diagnosis and Treatment cares for a high volume of monochorionic twin pregnancies each year. Since 1995, more than 4,745 complicated multiple gestation pregnancies have been referred to the Center for Fetal Diagnosis and Treatment, including 2,522 referrals for TTTS.
This experience helps to continuously expand our understanding of these complex pregnancies and our ability to differentiate between conditions such as TTTS, selective fetal growth restriction (sFGR), twin anemia polycythemia sequence (TAPS), and twin reversed arterial perfusion (TRAP) sequence.

Tour our Fetal Center
The Wood Center for Fetal Diagnosis and Treatment has cared for many families and will help you through your journey, too.

What to expect
From the moment of referral through delivery and postnatal care, your family can expect a supportive experience when you come to us with a diagnosis of a birth defect.
Resources to help
Twin-Twin Transfusion Syndrome (TTTS) Resources
Richard D. Wood Jr. Center for Fetal Diagnosis and Treatment Resources
Learning your baby has a birth defect is a life-changing experience. We want you to know that you are not alone. To help you find answers to your questions, we've created this list of educational health resources.