The history of eosinophilic esophagitis (EoE) is short — the disease was first identified less than 30 years ago — and significant challenges remain. But, for the first time, there is hope of successful treatment for children with the disorder — even for those with stubborn, refractory disease.
EoE is a rare disorder, occurring in an estimated 1 in 1,500 children, and involves swelling and inflammation of the esophagus, and excessive levels of immune cells called eosinophils. The condition is often discovered in early childhood — though it can affect older children and adults — and is linked to feeding difficulties, failure to thrive, weight loss, vomiting and swallowing challenges.
“In the past few years, there’s been an explosion of knowledge about EoE — and in the number of cases of children with this disorder,” says Amanda Muir, MD, a pediatric gastroenterologist and active researcher at Children’s Hospital of Philadelphia. Muir and Kathryn E. Hamilton, PhD, are Co-directors of the Epithelial Biology Program and Gastrointestinal Epithelium Modeling (GEM) Program, both part of CHOP’s Division of Gastroenterology, Hepatology and Nutrition.

“Our work spans bench-to-bedside as we try to figure out what’s causing the inflammation in patients with EoE; the factors that may affect EoE in patients — including genetics and the microbiome; and determining why some kids respond to the first line of therapy, while others have refractory disease,” Dr. Muir says.
To better understand EoE and each child’s individual disease, clinicians at CHOP have been collecting biopsy specimens during diagnostic procedures. Researchers then use cells from the samples to create patient-derived organoids (PDOs) that replicate the form and function of the esophagus on a small scale. Researchers use these PDOs to study disease development and, in collaboration with clinicians, to test the effectiveness of new therapies that treat EoE in patients who have failed to improve with established treatments.
“We’re on the precipice of breakthrough discoveries for EoE,” Dr. Muir says. “While no therapy is perfect for every patient, we hope to be able to offer more patients with EoE a range of personalized therapies to improve both their symptoms and quality of life.”
EoE care from multiple perspectives
The Center for Pediatric Eosinophilic Disorders at CHOP currently treats 1,500 infants, children and teens with EoE — one of the largest clinical populations in the world. This large clinical population provides an extensive source of data as specialists from gastroenterology, allergy, nutrition and pathology collaborate to diagnose and treat patients, while physicians and scientists work to better understand the disorder and its causes, and find targeted new therapies to help children with intractable disease.
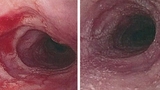
Children with feeding difficulties are often referred to CHOP after failed feeding therapy. “When children are young, its often hard to distinguish between picky eaters and those with EoE until an endoscopy is performed,” Dr. Muir says.
“We’re hoping to develop an algorithm to better identify EoE in younger children before feeding disorders become severe or they exhibit stiffening or narrowing of the esophagus,” Dr. Muir says.
New diagnostic tools
The only way to diagnose EoE is with a biopsy of the esophagus, taken during an endoscopy. Unfortunately, repeated endoscopic procedures are often required for diagnosis, to determine if the disease is worsening or if treatment is working. These endoscopies are necessary as long-standing disease can lead to extensive stiffening and narrowing of the esophagus. Until recently, there was no effective way to objectively measure and predict changes in esophageal stiffness or diameter.
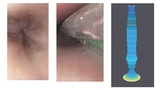
Enter the EndoFLIP® imaging system, an endoluminal functional luminal imaging probe, which was developed to measure the dimensions and function of organs like the esophagus (See related story on page 6.). Clinicians at CHOP have pioneered the use of EndoFLIP to measure esophageal diameter and pressure in real time. CHOP has performed 500-plus EndoFLIPs on pediatric patients in the past few years, and clinicians have discovered that children with EoE have increased stiffness and decreased esophageal distensibility compared to their healthy peers.
“We can use this information to help identify patients at risk of stricture and determine how aggressive treatment should be,” Dr. Muir says.
Another new diagnostic tool in use at CHOP is the transnasal endoscopy, introduced just before the COVID-19 pandemic hit the U.S. in 2020. In pediatric patients, there is often a disconnect between symptoms and biopsy results, making endoscopy with anesthesia a frequent occurrence for patients with suspected or confirmed EoE. Understanding disease activity without requiring anesthesia was not possible until transnasal endoscopy was introduced as an alternative for some older children. Scopes are inserted through the nasal passage, down through the throat and into the upper GI while children watch TV with virtual reality goggles. In 5-7 minutes, tissue samples can be taken, and scopes removed — offering an anesthesia-free alternative for some children and teens.
A third test recently studied at CHOP is an esophageal string test, Dr. Muir says. Patients swallow a capsule attached to a string that unravels as it travels from the patient’s mouth to their stomach. Then, the string is withdrawn and the part that was in the patient’s esophagus is sent to the lab to be tested for secretions. “This test will be able to tell us if their disease is still active or not,” Dr. Muir adds. Though scheduling details still need to be ironed out, this test is expected to be available to referring physicians by late spring.
Clinical trials
In addition to investigating diagnostic tools, researchers and clinicians at CHOP are also active participants in two drug trials that may prove beneficial for patients with eosinophilic esophagitis:
- Dupilumab (Dupixent®) is a twice monthly injectable medication used to block two sources of inflammation (interleukin 4 and interleukin 13) but does not suppress the immune system. This medication is generally used to treat eczema, asthma and nasal polyps.
- Lirentelimab (AK002), a monthly infusion therapy, uses humanized immunoglobulin monoclonal antibodies to target Siglec-8, an inhibitory receptor found on the surface of eosinophils and mast cells. Early research shows this therapy broadly inhibits mast cells and depletes eosinophils. Both studies are closed to new participants.

A third trial, led by Jonathan Spergel, MD, PhD, Chief of Food Allergy Center Frontier Program, is expected to launch soon to better understand how the immune system responds to allergens in patients with EoE. Clinicians and scientists hope current research efforts will lead to assays to help tailor therapies for patients with EoE, and shorten the time needed to find an effective therapeutic regimen for individual patients.
Upcoming studies
Successful research at CHOP has prompted further study into possible causes of EoE, including two studies about to start:
- A partnership with UNC Health in North Carolina to collect baby teeth from children with EoE, grind up the teeth, then test the individual samples for potential contributing factors to their disorder such as: how they were fed (breast milk vs. formula), their exposure to antibiotics in early childhood, and other factors that may help scientists determine who is at greater risk of developing EoE.
- A single-site study to expose patient-derived organoids to food additives and herbicides to evaluate the effect of these environmental triggers on cells and identify potential contributing factors in the development of EoE.
“Eosinophilic esophagitis is a dynamic and emerging field of study,” Dr. Muir says. “At CHOP, we have a clinical and bench science program that is ideal to study this disease and uncover novel therapeutics that will help our patients and ultimately, children with EoE around the world.”
Featured in this article
Specialties & Programs
The history of eosinophilic esophagitis (EoE) is short — the disease was first identified less than 30 years ago — and significant challenges remain. But, for the first time, there is hope of successful treatment for children with the disorder — even for those with stubborn, refractory disease.
EoE is a rare disorder, occurring in an estimated 1 in 1,500 children, and involves swelling and inflammation of the esophagus, and excessive levels of immune cells called eosinophils. The condition is often discovered in early childhood — though it can affect older children and adults — and is linked to feeding difficulties, failure to thrive, weight loss, vomiting and swallowing challenges.
“In the past few years, there’s been an explosion of knowledge about EoE — and in the number of cases of children with this disorder,” says Amanda Muir, MD, a pediatric gastroenterologist and active researcher at Children’s Hospital of Philadelphia. Muir and Kathryn E. Hamilton, PhD, are Co-directors of the Epithelial Biology Program and Gastrointestinal Epithelium Modeling (GEM) Program, both part of CHOP’s Division of Gastroenterology, Hepatology and Nutrition.

“Our work spans bench-to-bedside as we try to figure out what’s causing the inflammation in patients with EoE; the factors that may affect EoE in patients — including genetics and the microbiome; and determining why some kids respond to the first line of therapy, while others have refractory disease,” Dr. Muir says.
To better understand EoE and each child’s individual disease, clinicians at CHOP have been collecting biopsy specimens during diagnostic procedures. Researchers then use cells from the samples to create patient-derived organoids (PDOs) that replicate the form and function of the esophagus on a small scale. Researchers use these PDOs to study disease development and, in collaboration with clinicians, to test the effectiveness of new therapies that treat EoE in patients who have failed to improve with established treatments.
“We’re on the precipice of breakthrough discoveries for EoE,” Dr. Muir says. “While no therapy is perfect for every patient, we hope to be able to offer more patients with EoE a range of personalized therapies to improve both their symptoms and quality of life.”
EoE care from multiple perspectives
The Center for Pediatric Eosinophilic Disorders at CHOP currently treats 1,500 infants, children and teens with EoE — one of the largest clinical populations in the world. This large clinical population provides an extensive source of data as specialists from gastroenterology, allergy, nutrition and pathology collaborate to diagnose and treat patients, while physicians and scientists work to better understand the disorder and its causes, and find targeted new therapies to help children with intractable disease.

Children with feeding difficulties are often referred to CHOP after failed feeding therapy. “When children are young, its often hard to distinguish between picky eaters and those with EoE until an endoscopy is performed,” Dr. Muir says.
“We’re hoping to develop an algorithm to better identify EoE in younger children before feeding disorders become severe or they exhibit stiffening or narrowing of the esophagus,” Dr. Muir says.
New diagnostic tools
The only way to diagnose EoE is with a biopsy of the esophagus, taken during an endoscopy. Unfortunately, repeated endoscopic procedures are often required for diagnosis, to determine if the disease is worsening or if treatment is working. These endoscopies are necessary as long-standing disease can lead to extensive stiffening and narrowing of the esophagus. Until recently, there was no effective way to objectively measure and predict changes in esophageal stiffness or diameter.

Enter the EndoFLIP® imaging system, an endoluminal functional luminal imaging probe, which was developed to measure the dimensions and function of organs like the esophagus (See related story on page 6.). Clinicians at CHOP have pioneered the use of EndoFLIP to measure esophageal diameter and pressure in real time. CHOP has performed 500-plus EndoFLIPs on pediatric patients in the past few years, and clinicians have discovered that children with EoE have increased stiffness and decreased esophageal distensibility compared to their healthy peers.
“We can use this information to help identify patients at risk of stricture and determine how aggressive treatment should be,” Dr. Muir says.
Another new diagnostic tool in use at CHOP is the transnasal endoscopy, introduced just before the COVID-19 pandemic hit the U.S. in 2020. In pediatric patients, there is often a disconnect between symptoms and biopsy results, making endoscopy with anesthesia a frequent occurrence for patients with suspected or confirmed EoE. Understanding disease activity without requiring anesthesia was not possible until transnasal endoscopy was introduced as an alternative for some older children. Scopes are inserted through the nasal passage, down through the throat and into the upper GI while children watch TV with virtual reality goggles. In 5-7 minutes, tissue samples can be taken, and scopes removed — offering an anesthesia-free alternative for some children and teens.
A third test recently studied at CHOP is an esophageal string test, Dr. Muir says. Patients swallow a capsule attached to a string that unravels as it travels from the patient’s mouth to their stomach. Then, the string is withdrawn and the part that was in the patient’s esophagus is sent to the lab to be tested for secretions. “This test will be able to tell us if their disease is still active or not,” Dr. Muir adds. Though scheduling details still need to be ironed out, this test is expected to be available to referring physicians by late spring.
Clinical trials
In addition to investigating diagnostic tools, researchers and clinicians at CHOP are also active participants in two drug trials that may prove beneficial for patients with eosinophilic esophagitis:
- Dupilumab (Dupixent®) is a twice monthly injectable medication used to block two sources of inflammation (interleukin 4 and interleukin 13) but does not suppress the immune system. This medication is generally used to treat eczema, asthma and nasal polyps.
- Lirentelimab (AK002), a monthly infusion therapy, uses humanized immunoglobulin monoclonal antibodies to target Siglec-8, an inhibitory receptor found on the surface of eosinophils and mast cells. Early research shows this therapy broadly inhibits mast cells and depletes eosinophils. Both studies are closed to new participants.

A third trial, led by Jonathan Spergel, MD, PhD, Chief of Food Allergy Center Frontier Program, is expected to launch soon to better understand how the immune system responds to allergens in patients with EoE. Clinicians and scientists hope current research efforts will lead to assays to help tailor therapies for patients with EoE, and shorten the time needed to find an effective therapeutic regimen for individual patients.
Upcoming studies
Successful research at CHOP has prompted further study into possible causes of EoE, including two studies about to start:
- A partnership with UNC Health in North Carolina to collect baby teeth from children with EoE, grind up the teeth, then test the individual samples for potential contributing factors to their disorder such as: how they were fed (breast milk vs. formula), their exposure to antibiotics in early childhood, and other factors that may help scientists determine who is at greater risk of developing EoE.
- A single-site study to expose patient-derived organoids to food additives and herbicides to evaluate the effect of these environmental triggers on cells and identify potential contributing factors in the development of EoE.
“Eosinophilic esophagitis is a dynamic and emerging field of study,” Dr. Muir says. “At CHOP, we have a clinical and bench science program that is ideal to study this disease and uncover novel therapeutics that will help our patients and ultimately, children with EoE around the world.”
Contact us
Division of Gastroenterology, Hepatology and Nutrition