What is tricuspid atresia?
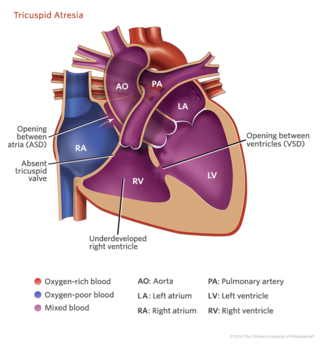
The tricuspid valve is the opening between the right upper chamber of the heart (the right atrium) and the right lower chamber of the heart (right ventricle). (In a normal heart, the right side receives blood that is low in oxygen from the body’s veins. It pumps the oxygen-poor blood to the lungs to receive oxygen. The oxygen-rich blood then returns from the lungs to the left side of the heart, which pumps the blood to the body.
A heart with tricuspid atresia is poorly developed on the right side, including:
- No tricuspid valve
- A right ventricle that is smaller than normal, also called “hypoplastic”
- A hole between the right atrium and left atrium (atrial septal defect) that allows oxygen-poor and oxygen-rich blood to mix inside of the heart
Most children with tricuspid atresia also have a ventricular septal defect, which is a hole between the right ventricle and left ventricle.
Tricuspid atresia in children is often linked with pulmonary stenosis, which is a narrowing of the pulmonary valve. Tricuspid atresia can also be linked with transposition of the great arteries, which is when the aorta (the large artery that carries oxygen-rich blood to the body) and the pulmonary artery (the large artery that carries oxygen-poor blood from the heart to the lungs) are connected to the heart abnormally.
Tricuspid atresia is a single-ventricle defect, because the heart has only one functioning ventricle (the left ventricle).
Signs and symptoms of tricuspid atresia
Tricuspid atresia symptoms in children include:
- Blue or purple tint to lips, skin and nails (cyanosis)
- Heart murmur, or abnormal heart sound when a doctor listens with a stethoscope
- Shortness of breath
- Difficulty feeding
- Poor weight gain
- Fatigue
- Abnormally shaped fingertips, called “clubbing”
Testing and diagnosis of tricuspid atresia
Tricuspid atresia may be diagnosed before birth, with a special kind of ultrasound called a fetal echocardiogram. Our Fetal Heart Program leads the nation in the evaluation, diagnosis and treatment of fetal heart disease before birth. Our experts will monitor both you and your baby throughout your pregnancy and create a plan for labor and care for your newborn after birth.
Usually, tricuspid atresia is diagnosed a few hours or days after a baby is born. Pediatricians from other hospitals often refer newborns to the Cardiac Center when a baby shows signs that may indicate a heart problem.
Diagnosing tricuspid atresia may require some or all of these tests:
- Electrocardiogram (EKG or ECG), which is a record of the electrical activity of the heart
- Echocardiogram (also called echo or ultrasound), which is when sound waves create an image of the heart
- Chest X-ray
- Blood tests
Sometimes, cardiac catheterization will be required. A thin, flexible tube (catheter) is inserted through a vein or artery in the leg and into the heart to provide detailed information about the structure and function of the heart and lungs.
Treatment for tricuspid atresia
If your baby is born with tricuspid atresia, they will be admitted to the Evelyn and Daniel M. Tabas Cardiac Intensive Care Unit (CICU), where they will receive round-the-clock attention from a team of dedicated cardiac critical care medicine specialists. Your baby may require oxygen and a medication called prostaglandin, to keep enough oxygen in their blood.
Prostaglandin is an IV (intravenous) medication that keeps the connection between the pulmonary artery and the aorta open. This connection, called the patent ductus arteriosus (PDA), is open in the fetus, and closes soon after birth. When the PDA closes, some babies with tricuspid atresia turn blue. An infusion of prostaglandin is a lifesaving intervention that can re-open the PDA. Not all babies with tricuspid atresia require prostaglandin.
It is not unusual for babies to have trouble breathing or stop breathing for short periods (apnea) while on prostaglandin infusion. If your baby struggles to breathe, they may need to be placed on a breathing machine or ventilator.
To repair many types of single ventricle defects, including tricuspid atresia, surgeons perform a series of open-heart procedures over a period of years. This is called “staged reconstruction.”
Blalock-Taussig-Thomas shunt or Ductal Stent
Babies who need prostaglandin to keep their oxygen levels stable will need surgery shortly after birth. The first surgery involves placing a “shunt,” which is a tube that connects a branch of the aorta to the pulmonary artery, taking over the job of the PDA. This surgery is called the Blalock-Taussig-Thomas (or BTT) shunt.
Many babies with tricuspid atresia are healthy enough to go home soon after birth. However, some may need the BTT shunt a few weeks later if their oxygen levels drop.
Instead of having a surgical stunt, some babies may have a ductal stunt placed. This means putting a small tube (stent) into the ductus arteriosus, the blood vessel that connects the pulmonary artery to the aorta. The vessel is open in the fetus but closes after birth. The stent keeps it open, allowing more blood to flow to the lungs.
Some babies with tricuspid atresia are too “pink” (meaning their oxygen levels are too high because of too much blood flow to the lungs). These babies need an operation called “pulmonary artery banding” to narrow the pulmonary artery and control blood flow to the lungs.
Babies who have tricuspid atresia and transposition of the great arteries may need the Norwood procedure.
Glenn
The second operation, called the Glenn operation, usually happens within six months of a child’s birth. During this surgery, the superior vena cava — the large vein attached to the heart that returns deoxygenated blood from the upper half of the body — is closed off or disconnected from the heart and instead attached to the pulmonary artery. The surgeon will also close the BTT shunt or remove the ductal stent. After the Glenn, deoxygenated blood from the upper body goes directly to the lungs without passing through the heart.
Fontan
The third operation, called the Fontan, usually happens when a child is 18 months to 3 years old. During this surgery, blood from the inferior vena cava — the large vein that returns deoxygenated blood to the heart from the lower half of the body — is diverted directly to the pulmonary artery. This means that deoxygenated blood from the whole body now goes to the lungs without passing through the heart. This is called Fontan circulation.
Your Cardiac Center team will explain staged reconstruction to you in greater detail based on your child’s individual anatomy.
Outlook for tricuspid atresia
Because of enormous strides in medicine and technology, today many children born with tricuspid atresia go on to lead healthy, productive lives as adults.
Follow-up care
Through age 18
Though early outcomes for children with single ventricle heart defects after staged reconstruction have greatly improved over the years, the time between the Norwood procedure and the Glenn operation is a very vulnerable time for infants. Our Infant Single Ventricle Monitoring and Management Program provides care and monitoring for babies with single ventricle heart defects between two necessary heart repair procedures.
Children with tricuspid atresia require life-long care by a cardiologist. Many remain on medications for life. Additional surgeries may be required.
In addition, children with complex congenital heart defects who have had open heart surgery as infants are at a higher risk for neurodevelopmental issues when compared to children without congenital heart defects. CHOP’s Cardiac Kids Development Follow-up Program provides evaluation, screening and clinical care for children with complex congenital heart disease who are at risk for neurodevelopmental problems.
As patients with Fontan circulation get older, doctors are recognizing that, while some do fine, many experience complications. These complications can include lung, liver and gastrointestinal diseases. Our Single Ventricle Survivorship Clinic, part of the Fontan Rehabilitation, Wellness, Activity and Resilience Development (FORWARD) Program specializes in the care of these patients and is researching improved treatments for them.
Into adulthood
Our pediatric cardiologists follow patients until they are young adults. The Philadelphia Adult Congenital Heart Center, a joint program of CHOP and Penn Medicine, meets the unique needs of adults who were born with heart defects. At CHOP’s Cardiac Center, we support our adolescent and young adult patients through a smooth and seamless transition to adult cardiology care at the Philadelphia Adult Congenital Heart Center or, if desired, to a cardiologist with congenital heart disease expertise in another location.

Why Choose Us
Our specialists are leading the way in the diagnosis, treatment, and research of congenital and acquired heart conditions.

Patient Outcomes at the Cardiac Center
Children’s Hospital of Philadelphia's pediatric heart surgery survival rates are among the best in the nation.
Resources to help
Cardiac Center Resources
We know that caring for a child with a heart condition can be stressful. To help you find answers to your questions – either before or after visiting the Cardiac Center – we’ve created this list of educational health resources.
Reviewed by Jenna Heichel, MSN, BSN, CRNP
Reviewed on 08/08/2024
